
Latest News from VivaZome
Check out our latest news and media releases
Our mission
To be a global leader in providing novel, valuable and safe exosome-derived therapies for debilitating and life-threatening diseases
Our focus is on developing therapies for debilitating and/or life-threatening disorders where current treatment are inadequate, have low efficacy or are poorly tolerated. We look to established pre-clinical disease models that have potential for VivaZome therapy to provide a clinically dramatic treatment effect.
With a global orientation from the outset our mission is to take the VivaZome therapies to the world’s major markets. Critical to this is early engagement with regulators in major jurisdictions and securing global or regional partners to support clinical studies, access and sales in major markets.
Identify and secure potential cell sources for exosomes for the target disorder. Characterisation and definition of the exosomes. Conduct of small-scale pilot clinical studies to demonstrate safety and, where possible, provide efficacy signals. Undertaking multi-national multi-centre clinical studies to support product registration and reimbursement.
Development of a scale-able, validated, cost-efficient GMP manufacturing process, with fully-integrated quality systems. This may include licensing of relevant technologies and/or joint projects with major technology providers.
Raising investor and grant capital to fund the development and scientific work. Ongoing scientific studies to enhance the clinical utility of the therapies and further strengthen the international IP position.
Project management to ensure all activities are closely managed for cost and timing and milestones are achieved.
Exosome technologies
Exosome therapy is a relatively new concept, with both challenges and significant opportunities
Why exosomes?
Exosomes are nanosized vesicles (50-200nm) released naturally by cells. They are important effectors of cell communication. A key mechanism by which cell therapies work is via release and uptake of exosomes.
Some of the properties of exosomes are:
- they contain unique mix of molecular cargo (including miRNA, proteins, peptides, lipids) that underpin their biological effect
- they have homing capabilities that allow them to travel to the site of injury or disease
- their immunomodulatory properties helps normalise the immune system
- being low/non-immunogenic they are not rejected by the body
- they have other bioactive properties based on parent cell e.g. angiogenesis, anti-fibrotic, regenerative, etc.
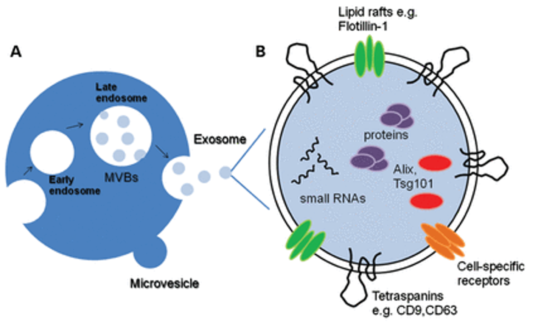
A: Cell
B: Exosome
Meet our board and management
Over 120 years of combined biotech, cell biology and pharmaceutical experience

Ian Nisbet
Ian has over 30 years biologicals and biotech experience in senior management, corporate advisory and company director roles.

Craig Newton
Craig has more than 30 years experience in the medical device, pharmaceutical and biotech sectors in Australia, Asia and Europe.

David Haylock
David has over 35 years experience in cell biology, cell therapies and operational management with senior scientific leadership roles.

Xenia Sango
Xenia is an experienced leader in healthcare with over 30 years in senior operational, commercialisation, clinical, regulatory roles.
Contact us
Directors
Ian Nisbet PhD
Email: ian.nisbet@vivazome.com
Tel: +61 (0) 431 709 121


